We’ve all been part of initiatives that chart a new path and make an impact. But when we apply the word “process” to these undertakings, some shut down and perceive it as a “box-ticking” exercise. Done well, however, process does not thwart creativity — it liberates innovation, facilitates strategic thinking, and drives cross-functional collaboration.
Here’s how…
The Strategic Planning Hierarchy
Strategic planning within a company must be interconnected, with a clear hierarchy that aligns every effort with the overarching vision and mission. As plans cascade down the organization, they increase in specificity from corporate-level decisions on therapeutic areas and platforms to asset-level, tactical execution.
While the exact structure varies by company maturity and design, an illustrative planning framework may look like this:

(Click image to expand)
It is important for the team to consistently align around a shared lexicon, with clear definitions of plan scope approved by leadership. Establishing a common vocabulary and outline can eliminate wasted effort and miscommunication during the planning process.
Communicating a planning calendar with clear expectations is also critical. This gives plan contributors sufficient time to prepare and gather valuable stakeholder input, ensuring the plans are insightful and representative.
Commercialization Planning and Timing
Once the broader corporate plans are in place, the commercialization plan — describing how a product will reach and benefit patients — comes next. It is, in effect, the bridge between scientific innovation and patient impact.
The optimal time for creation of the first commercialization plan for a development asset is three years (or more) before launch — typically during mid-late Phase 2 — as it informs key elements of the P3 trial and plan, and provides insight into the resources and budget.
This is the stage at which commercialization investment starts to ramp up; the plan provides a shared foundation for strategic decision-making at the market level. Also, if a company hopes to partner its asset, knowing the scope of commercialization required will be essential in future negotiations.
The commercialization planning process should be updated annually and aligned with other planning processes, such as long-range planning and budgeting (through product launch and beyond), to ensure the strategy is continuously refreshed in response to changing market dynamics and internal goals.
While the process is usually led by commercial (sometimes by program leadership), it is truly a cross-functional exercise involving all roles in product commercialization. While the plan roadmap is vital as a reference and communication tool, engagement across various functions is often the most valuable aspect of commercialization planning.
Overall, the planning process provides a forum and structure for collaborative strategic discussions and turns creative thinking into tangible action. A structured process frees teams from scrambling to figure out how to plan, and instead removes friction and provides space for focusing on what needs paying attention to. We have witnessed many “lightbulb moments” that have made a critical difference to how a product is commercialized.
Elements of the Commercialization Process
A successful process ensures the right questions are answered at the right time and should begin with an external perspective.
A deep assessment of the market — treatment and diagnostic trends, competition, regulation, reimbursement, etc. — is essential for informing the strategy, ensuring the product’s value proposition is meaningful to customers, and identifying challenges early. A comprehensive understanding of the external environment and its impact on product and patient access is increasingly important as life sciences grows in complexity, with new stakeholders, policy changes, and technological innovation.
A robust situation analysis can be aided by internally vetted AI tools, thanks to their skill in synthesizing vast amounts of information (e.g., published literature, conference abstracts, analyst reports, competitive intelligence), identifying patterns in patient journeys, surfacing unmet and as yet unarticulated needs, and flagging competitive signals early. That said, the outputs generated by AI are no replacement for human thinking and judgment and should only be used to enhance the team’s knowledge and view of the market.
The situation analysis provides context for product strategy development and shared understanding, and is ideally developed through a series of discussions and workshops. Once there is alignment around the strategy, functional tactical plans and associated budgets should ladder up to strategic objectives.
Technology can help facilitate the cross-functional strategy development and tactical planning using platforms (Corval, Smartsheet, Miro Boards) that foster collaborative brainstorming, flag inconsistencies, and surface resource conflicts.
Tools that sit across the planning process and connect inputs from different teams in real time represent a significant opportunity to improve plan coherence and reduce costly misalignments that emerge late in the planning cycle.
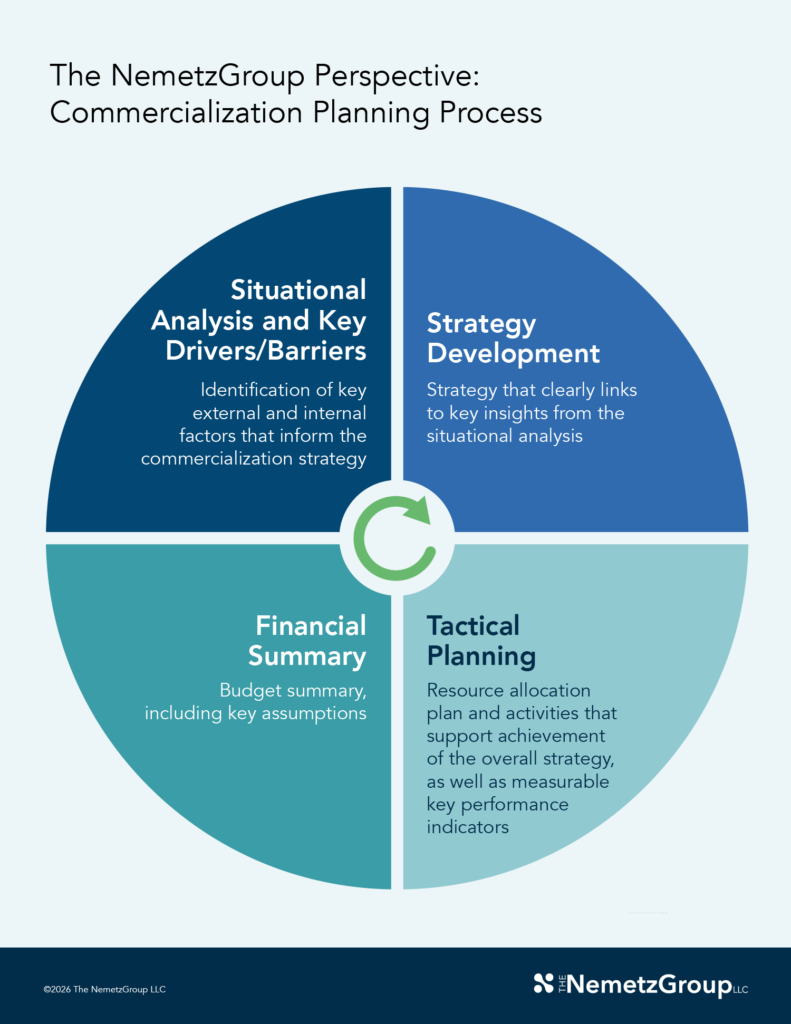
(Click image to expand)
One Brand Planning Process Does Not Fit All
Not all companies will have the time, resources, or appetite to implement a comprehensive strategic planning process. Therefore, the process should be calibrated based on the company’s situation and culture. Nevertheless, we strongly recommend that some variation of the commercialization planning process be undertaken for all pre-commercial and in-line (on the market) products/portfolios.
Companies that choose a less formal approach are often plagued by “strategic drift,” where functions are not aligned, resulting in the implementation of tactics that may be familiar, but not necessarily fit for purpose. This leads to a misallocation of resources, where investment decisions are made reactively, with no clear rationale connecting spend to strategy. Ultimately, the internal disconnect manifests itself externally, as customer engagement is not purposeful or coordinated.
Don’t Shortchange This Step
The commercialization planning process is a valuable opportunity. It results in a shared vision and understanding of the product strategy and the cross-functional initiatives that will be implemented to make that strategy a reality.
Companies that get this right use process as a competitive advantage and are well prepared for navigating the complexity and uncertainty of biopharma commercialization.
We’ve partnered with clients at every stage to build brand planning processes that work. They are collaborative, stage-appropriate, and designed to create alignment. If you’re trying to figure out what “right-sized” looks like for brand planning at your organization, we’d be happy to share what we’ve learned along the way. Get in touch.
-

Colleen has consulting and biopharma experience in marketing, strategy development, operations, and project management within the life sciences and high-tech industries. Colleen’s keen analytical skills, collaborative problem-solving approach, and broad exposure across therapeutic areas enable an interdisciplinary approach to the key questions of commercial and brand strategy.
Read Colleen's full bio, here.